
I’ve just spent a week interpreting and sight translating for a clinical trial inspection at a hospital in Madrid. While I can’t share many of the details, here’s an anonymised review of the week and how I prepared for it.
My day-to-day work is desk-based – I translate clinical trial documentation, research articles and individual patient’s medical reports. So a daily commute to a clinical trial site, communicating with the research team and liaising with the inspector was quite different from my usual routine.
Audits and inspections
It’s not the first time I’ve been hired in this capacity. Last year I assisted at a pharmaceutical distribution plant audit in Madrid where a UK auditor was reviewing compliance with Good Manufacturing Practices and needed support with sight translation and communicating with the local team.
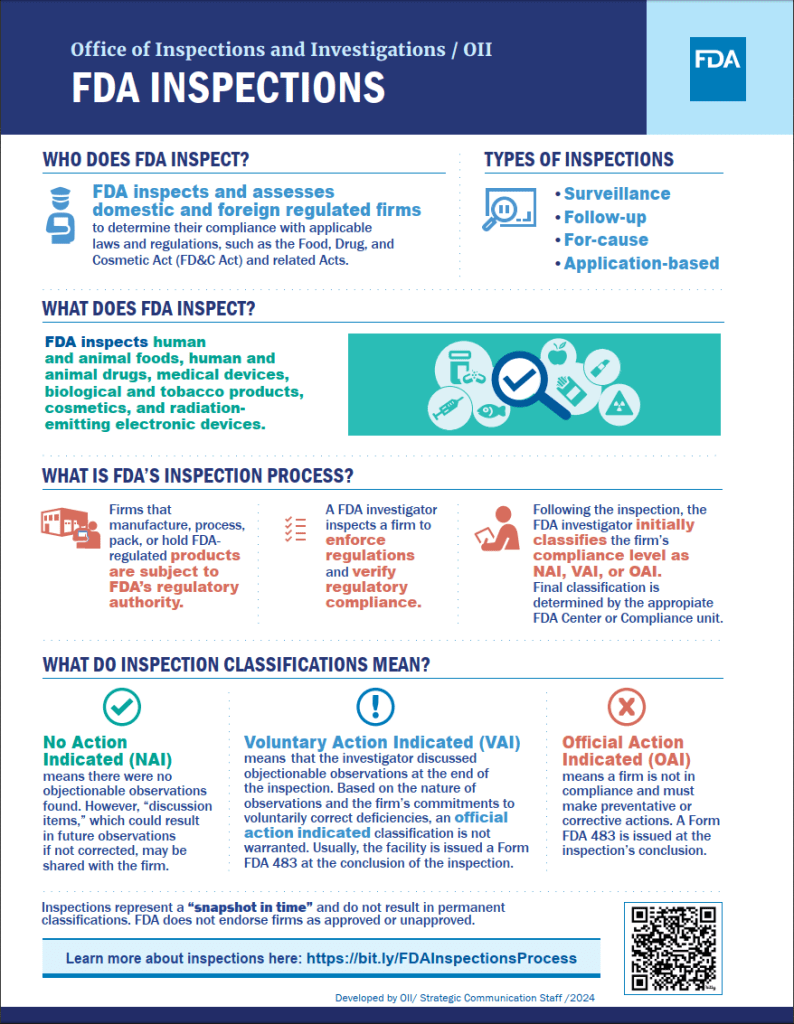
This time, the US Food and Drug Administration (FDA) announced it was going to carry out a routine inspection of an international clinical trial site in Madrid. The purpose of this type of inspection is to ensure that participants’ rights, safety and welfare are being protected, that the principal investigator is conducting the trial as per protocol and in compliance with regulatory requirements, and that clinical trial records match the on-site source documentation (medical records, ethics committee approvals, lab results, ECG reports, etc.). All with a view to granting marketing authorisation for the investigational medicinal product at the end of the trial in the event of a successful trial outcome.
Confidentiality
These inspections take place on site as records with personal data can be accessed only by authorised staff under strict conditions and after all consents and agreements have been signed. Some files and folders can then be shared and documented through a request log and others are restricted to over-the-shoulder access only.
Preparation
With the job booked over a month in advance, I spent the weeks leading up to the inspection brushing up on my note-taking skills, creating symbols for commonly used terms, and reading the 88-page trial protocol and other trial-related documents. We had a preparatory meeting two weeks beforehand to set expectations for the investigators, study coordinators and CRAs, and, in my case, to note down all the brand names used for the various platforms.
Back at home, I ran through standard questions that inspectors ask at these visits, and even recorded potential answers in Spanish, to practise listening, taking notes and speaking them back in English. I created an Anki flash card deck to drill domain-specific terms and create a set of purpose-designed symbols.
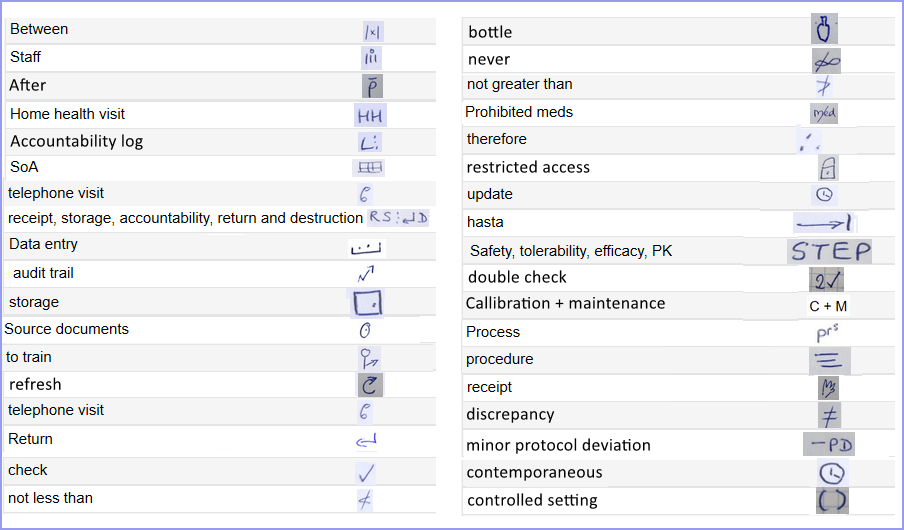
My symbols included pictograms for symptoms specific to the disease in question; Latin abbreviations (e.g. ā for before, p̄ for after); acronyms for common word combinations (e.g. STEP for “safety, tolerability, efficacy and pharmacokinetics”) and symbols for commonly used words (e.g. storage, restricted access, source documents). As when you learn anything new, I built up my symbol repository gradually, increasing by small steps, until I had a solid bank of just over 100 squiggles, probably legible only to me.
At the hospital
On the ground, the symbols were put to good use. Although most staff members had excellent English, I supported a study pharmacist and a study coordinator whose English wasn’t strong enough under the pressure of the inspection, when every word counts.
Their spoken Spanish, peppered with English medical terms, kept me on my toes: “el screening”, “un SAE”, “una note-to-file”, “el status” and “el delegation”. Quite different from formal written Spanish, which rarely uses English terms. And when communicating with the FDA inspector, I noticed several differences between US and UK English, such as schedule of activities (visits calendar), IRB (REC), participant welfare (participant well-being), binder (folder) and home health visits (at-home visits) for the US (UK), respectively.

As part of the inspection we also did a pharmacy walk-through to check drug accountability and chain of custody from delivery to destruction. And I spent hours scouring Spanish source documents housed in cumbersome participant and site folders, cross-checking them against the English study records of adverse events, permitted concomitant medications and study eligibility criteria, to flag any potential discrepancies between source and study records.
Wrap-up
Although I can’t disclose the outcome of the inspection, the visit went smoothly because the research team had prepared well, the inspector was efficient and pleasant, and communication flowed easily between the two parties. Human interaction at its best.
Feature photo by Julia Koblitz on Unsplash.
FDA infographic


Really interesting post, Emma! One of the things I found most striking was how clearly it illustrates the gap between documented processes and what actually happens “on the ground” during real-world workflows and inspections.
Thank you for sharing such a valuable behind-the-scenes perspective!
Oustanding, Emma! What an interesting task you had and how well you explained your procedure! Congrats!